
Few case reports from the field of cardiovascular surgery were published pointing out this problem. The use of AA prior to interventions with intended heparin anticoagulation has been reported to cause unresponsiveness to heparin. UFH is administered during open vascular and endovascular procedures to reduce the risk of thromboembolic events. In the absence of such a randomized controlled trial, safety and efficacy are being questioned by different groups of health care professionals, especially when considering the high cost of therapy with AA. Results of the ongoing ANNEXA-I trial (NCT03661528), a randomized controlled trial evaluating the efficacy and safety of AA versus usual care in patients with ICH on FXa inhibitors, are excitedly awaited. 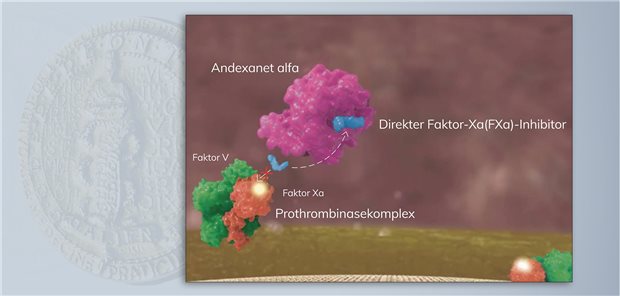
The authors conclude that AA reduced anti-FXa activity in FXa-inhibitor-treated patients with traumatic as well as spontaneous ICH with high rates of hemostatic efficacy. Just recently, the post hoc ANNEXA-4 substudy on hemostatic efficacy and anti-FXa reversal in intracranial hemorrhage (ICH) was published. 
This was followed by the ANNEXA-4 study published in 2019 on 352 patients who had acute major bleeding within 18 h after administration of an FXa inhibitor. Both studies analyzed anti-FXa activity in healthy volunteers receiving either apixaban (ANNEXA-A) or rivaroxaban (ANNEXA-R). Safety and efficacy were evaluated in two prospective randomized placebo-controlled studies. Andexanet alfa (AA) is the only reversal agent approved by the European Medicines Agency (EMA) and US Food and Drug Administration for oral direct factor Xa (FXa) inhibitors rivaroxaban and apixaban in situations when reversal of anticoagulation is needed because of life-threatening or uncontrolled bleeding. With a growing number of patients treated with direct oral anticoagulants, general interest in reversal agents in patients with hemorrhagic stroke is rising.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
